Move!plx® - A lemon verbena extract
Stress, Fatigue, Sleep and Sports Recovery
move!plx® is an aqueous lemon verbena extract containing phenylpropanoids that accelerate exercise recovery in athletes by reducing the risk of muscle soreness and injury and enhancing joint mobility.
move!plx® is also beneficial for sleep, mood and stress.
- Possesses anti-oxidant and anti-inflammatory properties
- Reduces markers associated with muscle damage
- Reduces muscle soreness
- Improves sleep and mood
- Improves joint mobility
move!plx® is available in France and in Benelux countries.
move!plx® is an aqueous extract of lemon verbena (Aloysia citrodoria Palau) leaves and is manufactured in Spain. It contains phenylpropanoids such as verbascosides and flavonoids such as luteolin.
move!plx® reduces oxidative stress and muscle damage, and thus helps reduce inflammation. move!plx® thus helps to reduce muscle soreness, which in turn improves sleep and mood in athletes.
move!plx® was developed by Monteloeder in collaboration with the Institute of Molecular and Cellular Biology at the Miguel Hernandez University in Spain.
For more information, visit the website on MOVE!PLX®.

Funes et al. (2009) characterized move!plx® using high-performance liquid chromatography (HPLC). move!plx® contains 25% of verbascosides.
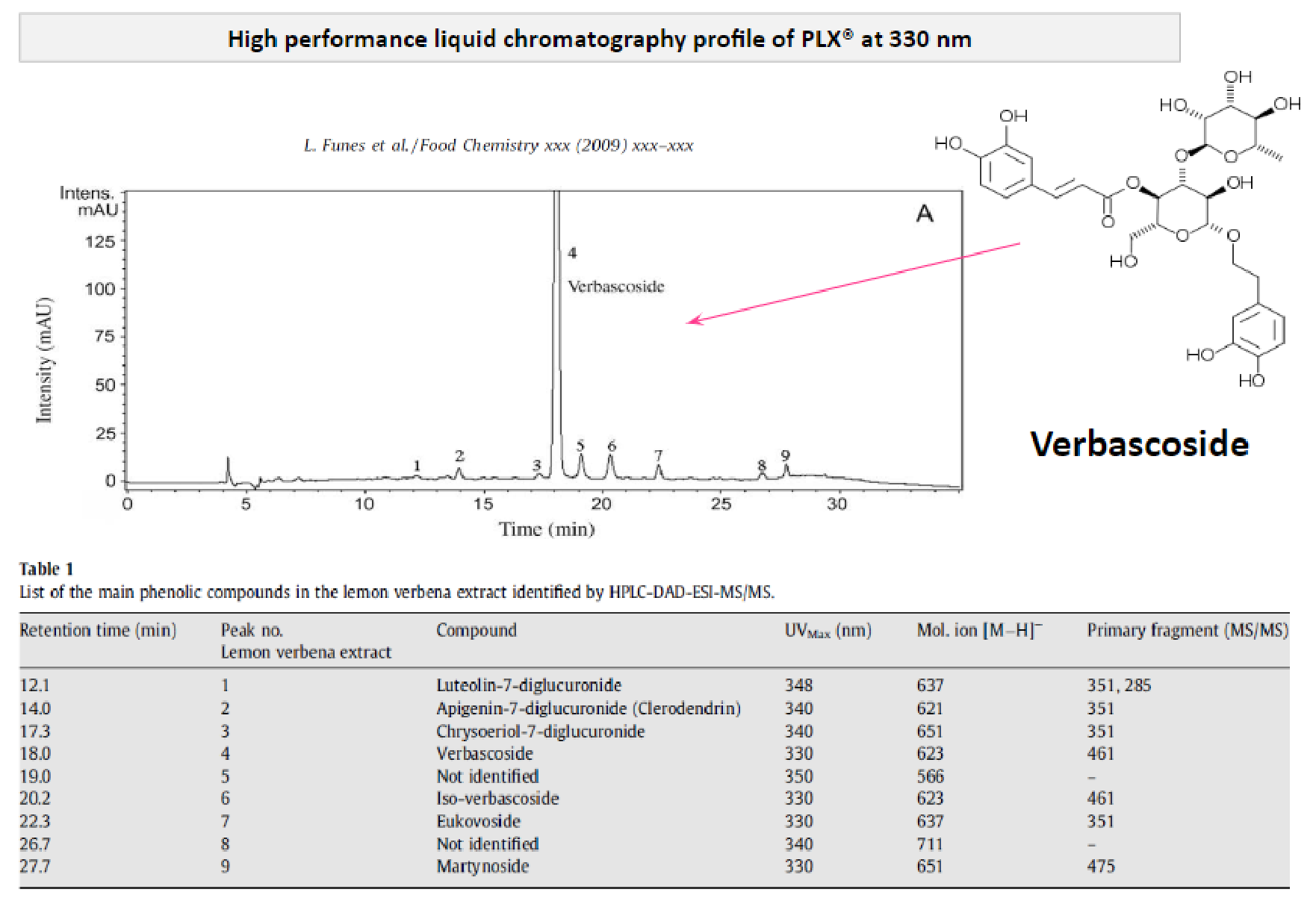
In addition, this study also showed a direct correlation between the anti-oxidative activity of the lemon verbena extract and the concentration in verbascosides (major bioactive ingredient in the extract).
Acute toxicity was assessed by administering an oral dose of 2000 mg/kg of lemon verbena extract to a test group of rats and results were compared with a control group, that did not receive any extract. No unwanted effects were observed after 15 days. Thus, move!plx® shows no toxicity below a massive dose of 2000 mg/kg.
Sources
- Funes L et al. “Correlation between plasma antioxidant capacity and verbascoside levels in rats after oral administration of lemon verbena extract”. Food Chemistry 2009; 117(4):589-98.
move!plx® is a lemon verbena extract that contains phenylpropanoids with many benefits on:
a. Reduces oxidative stress
b. Up-regulates glutathione reductase
c. Reduces markers of inflammation
d. Reduces markers of muscle damage
2. Sleep and stress : reduces muscle pain and improves sleep and mood
1. move!plx® and sport recovery
move!plx® showed results on muscle recovery following exercise, the reduction of muscle soreness.
Of the two pre-clinical and six clinical studies that provide evidence for the health benefits of move!plx®, four are presented here. The first three studies involve physiological markers of exercise recovery, one of which also includes assays of muscle soreness, sleep and mood. The fourth study addresses joint pain.
The protocol of the first three studies was identical. Two groups of students in Sports and Exercise Science at the Miguel Hernandez University in Spain underwent a physical training program consisting of a 90 min run (14.5 to 16.5 km) every other day for three weeks. One group received a supplement with a dose of 1200 mg of move!plx® at 10% verbascosides and the other group (control) received a placebo. The markers of anti-oxidative status, muscle soreness and inflammation were quantified and their differences were tested using an analysis of variance (ANOVA). The third study also studied psychological parameters such as mood.
a. Reduction of oxidative stress
In the first study, Carrera-Quintanar et al. (2010) investigated the anti-oxidative capacity of move!plx® on the oxidative status of plasma and blood cells (erythrocytes and lymphocytes) in 15 students who exercised three days a week (90 min, 14.5 to 16.5 km run) for three weeks. Following a randomized, double-blind protocol, eight students received a daily dose of 1200 mg of move!plx® (titered at 10% verbascosides) and seven students received a placebo. A two-way ANOVA on the factors “group” and “time” was used to test the differences between groups and between the beginning and the end of supplementation.
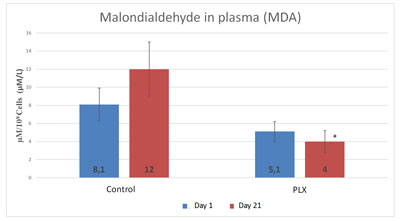
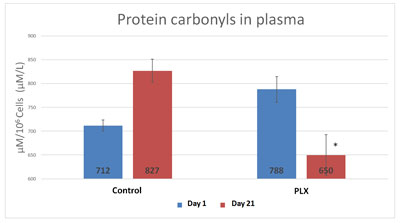
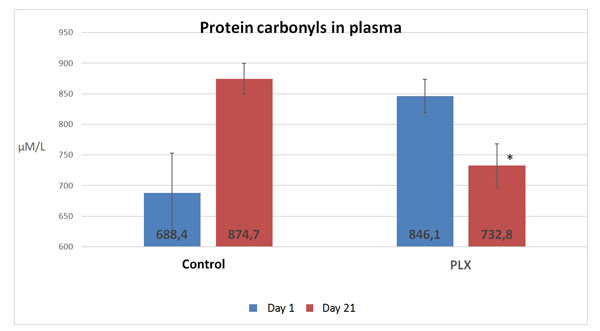
Blood samples were taken at 0 and 21 days and showed a significant decrease (p<0.05) in the markers associated with oxidative stress such as malondialdehyde (MDA) and protein carbonyls in the plasma.
According to this study, there was a significant difference in the concentration in MDA and protein carbonyls in the plasma between the group that received move!plx® and the group that did not after a 21 day training program, as illustrated in the figures below.
Likewise, in the second study (Funes et al., 2010) that measured the influence of move!plx® supplementation on the anti-oxidative response of neutrophils, markers associated with muscle damage and the cytokine profile of 15 students who followed a thrice weekly training program of 90 min consisting of a 14.5 to 16.5 km run for three weeks. In the randomized, double-blind protocol, eight students received a daily dose of 1200 mg of move!plx® (titered at 10% verbascosides) and seven students received a placebo.
MDA and protein carbonyls are makers of oxidative stress due to intensive exercise. They are the derivatives of the peroxidation of lipids and proteins that can compromise the proper functioning of leukocytes (chemotaxis, phagocytosis, etc.).
After 21 days, the level of MDA in the neutrophils was significantly lower (p=0.05) in the group that received move!plx® supplementation (EXT) than in the control group (PLB). Similar results were observed for protein carbonyls: there was a significant increase (p=0.08) in the control group but there was no significant difference over time in the group that received move!plx® supplementation.
MDA and protein carbonyls in neutrophils
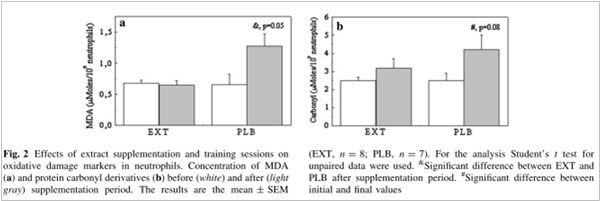
In the third study (Martinez-Rodriguez et al., 2015), the effect of move!plx® was measured on physiological parameters (MDA, protein carbonyls, γ-glutamyltransferase (GGT), aspartate aminotransferase (AST), myoglobin (Mb), myeloperoxidase (MPO)) in lymphocytes, neutrophils and erythrocytes [CE1] and also psychological parameters (POMS questionnaire, sleep quality and duration).
Using a randomized, double-blind protocol, eight students received a daily dose of 1200 mg of move!plx® (titered at 10% verbascosides) and seven students received a placebo. All followed the same training program with a 90 min run (14.5 to 16.5 km) every other day for three weeks.
Protein carbonyls and MDA were analyzed in the blood plasma, lymphocytes and neutrophils. In this study, only the activity of protein carbonyls in the plasma was significantly different (p<0.05) between the two groups.
In conclusion, move!plx® has a clear effect on oxidative stress markers and this effect was observed in all three clinical studies.
b. Up-regulation of glutathione reductase
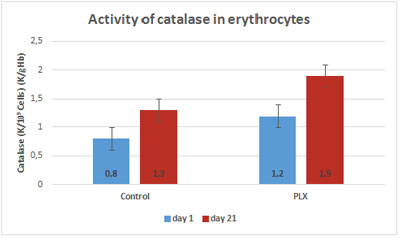
In the first study (Carrera-Quintanar et al., 2010), the three-week training program led to a significant increase in catalase, GSH reductase, GSH peroxidase and superoxide dismutase (SOD) in both groups. However, the activity of catalase and GSH reductase in the lymphocytes was significantly higher in the group that received move!plx® supplementation after exercising.
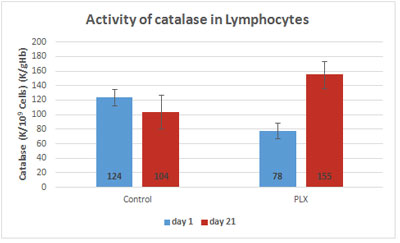
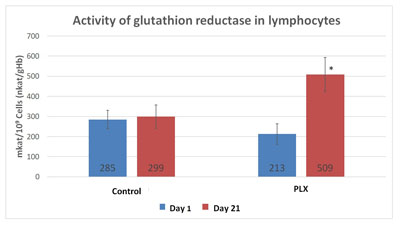
Given the importance of GSH-reductase in regenerating glutathione, the expression of the gene coding for glutathione reductase was measured in two groups using qRT-PCR, a technique for quantifying DNA. There were no differences in mRNA levels between the two groups, suggesting that regulation occurs at the protein level.
To better understand the changes observed in lymphocytes, the same enzymatic activities were measured in erythrocytes, cells without nuclei and thus whose regulation is not controlled by genes. As in the lymphocytes, the activity of GSH reductase strongly increased in the erythrocytes of the group that received move!plx® supplementation, again supporting the hypothesis that move!plx® regulates GSH reductase.
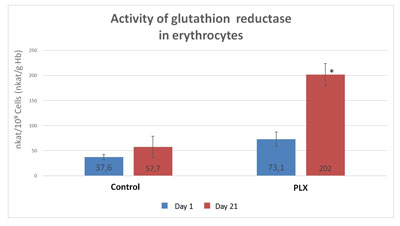
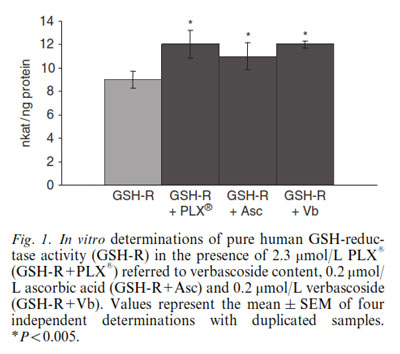
To explain the mechanism of GSH-reductase regulation, the enzyme was quantified using specific antibodies in lymphocytes in the two groups of students. No differences in the quantity of GSH-reductase was observed, suggesting that move!plx® modulates GSH-reductase activity rather than its quantity. To test this hypothesis, the activity of pure human GSH-reductase was measured in vitro in the absence and in the presence of move!plx®, purified verbascosides and ascorbic acid. The GSH-reductase activity was significantly higher (p<0.005) in the presence of move!plx®, verbascosides [CE1] and ascorbic acid, suggesting that move!plx® stimulates GSH activity through a redox mechanism.
This study won second place for the Spanish national award in sports medicine research.
Another study (Funes et al., 2010) measured glutathione reductase activity in neutrophils, which increased more in the supplemented group than in the control group. Again, this increase was not due to an increase in gene expression, suggesting that this activation acts at the post-transcriptional level.
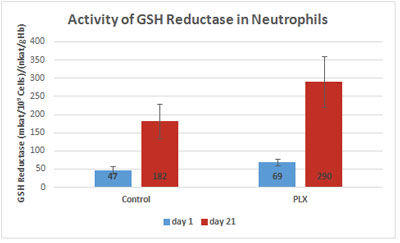
Therefore, move!plx® stimulates the natural anti-oxidant system through a redox effect on glutathione reductase.
c. Reduction in inflammation
In the Funes et al. (2010) study described above, there was a significant inhibitory effect (p<0.05) on myeloperoxidase (MPO) activity — a marker associated with inflammation— in the move!plx®-supplemented group compared with the control group. Several studies have shown that physical exercise favors the activation of MPO (enzyme that triggers the production of hypochlorous acid (HOCl), a powerful oxidant and early marker of inflammation) in neutrophils that can lead to excessive production of free radicals.
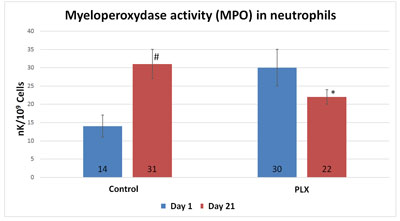
Another study also demonstrated the protective effect of move!plx® on neutrophils against oxidative stress and inflammation caused by exercise.
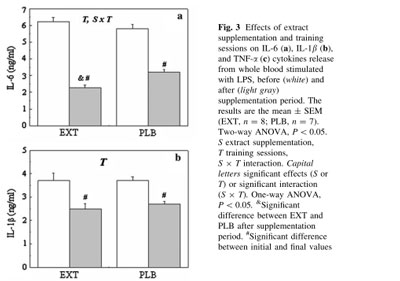
Moreover, the anti-inflammatory effect of move!plx® was assessed on cultured blood samples stimulated with a lipopolysaccharide (LPS), a pro-inflammatory molecule. Before and after the 21 day exercise program, concentrations of IL6, IL1B and TNF-alpha were assayed using an ELISA test.
Although IL6 was significantly reduced in both groups between the beginning and at the end of the training program, the decrease in IL6 was significantly higher (p<0.05) in the supplemented group than in the control group at day 21. Interleukin IL1B dropped significantly in each group but there was no significant difference between the two groups after 21 days.
In the third study, myeloperoxidase decreased significantly (p<0.05) (Alejandro Martinez-Rodriguez et al., 2015).
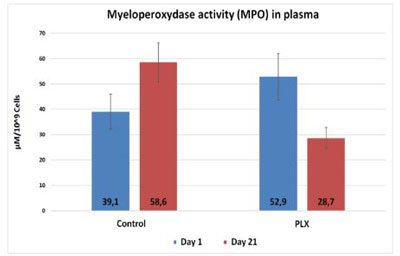
There is promising evidence that move!plx® has an effect on the markers of inflammation.
d. Reduction in markers of muscle damage
In the second study, the activity of alanine aminotransferase in the serum increased in the control group (+12.5%) whereas it remained stable in the group that received move!plx® supplementation. Similarly, the activity of aspartate aminotransferase was significantly reduced (-22%) in the group that received move!plx® supplementation in comparison with the control group. Finally, move!plx® supplementation led to a significant reduction (-31%) of gamma glutamyl transferase but no effect was observed in the control group.
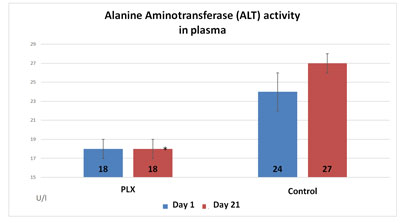
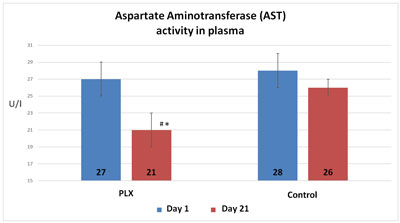
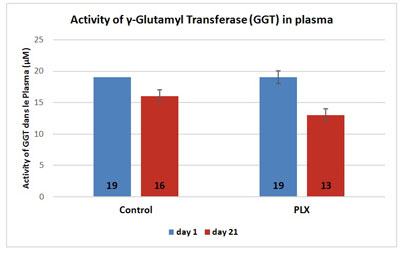
Thus, stability or decrease in aminotransferase activity indicates that move!plx® has a protective effect on muscle tissue.
In the third study, the group that received move!plx® supplementation showed plasma values that were significantly lower in aspartate aminotransferase and glutamic pyruvic transaminase than the control group. Similarly, the move!plx®-treated group showed plasma myoglobin (Mb) levels that were significantly lower (p<0.05) than in the control group. These results tend to show that move!plx® reduces the renewal of muscle tissue following exercise.
In conclusion, move!plx® seems to significantly reduce the markers of muscle damage due to intensive physical exercise.
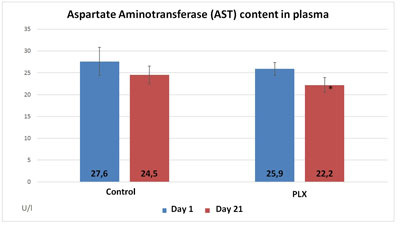
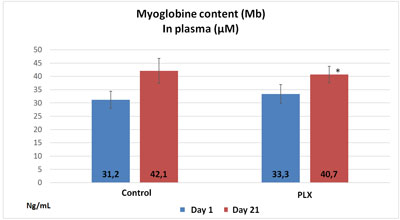
2. move!plx® and improved sleep, mood, well-being
Improvement of psychological parameters of sleep and mood
To evaluate the effect of move!plx® on exercise recovery, although it is necessary to measure biochemical parameters, psychological parameters are useful to obtain a full picture of the physical and mental state of athletes that consume move!plx® compared with the control group.
A Profile of Mood States (POMS) questionnaire administered in the third study assesses the emotional state of the participants through answers to questions on feelings of tension, depression, anger, vigor, fatigue and confusion and their sub-profiles. It was administered at the end of the three-week study. A five-item questionnaire on sleep duration and quality was administered every morning upon waking. Soreness was also recorded by participants.
The questionnaire on sleep shows that volunteers in the move!plx®-supplement group slept significantly longer (p<0.05, 20 minutes on average) than volunteers in the control group. Similarly, the number of nights during which volunteers woke up at least once was significantly higher in the control group. Finally, muscle soreness upon waking was slightly but significantly higher in the control group than in the move!plx-supplement group.
Therefore, the group that received move!plx® supplements enjoy more fitful sleep than the control group.
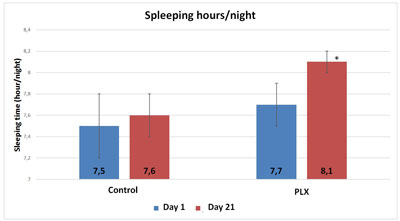
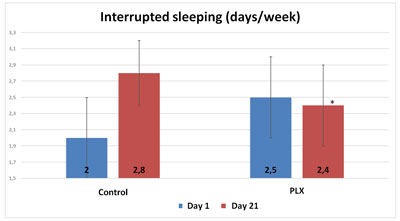
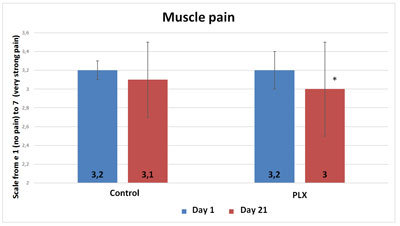
Also, although the total score of the POMS questionnaire did not show any significant differences between the two groups, three specific “mood profiles” of the questionnaire showed results that were significantly improved in the group that received move!plx® supplements. The mood profiles that showed differences involve anxiety, anger and fatigue.
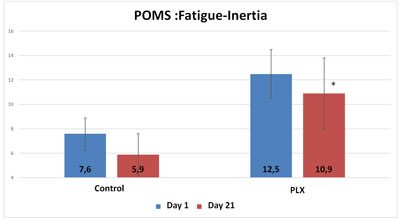
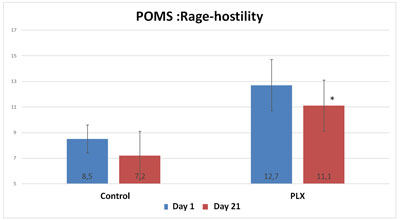
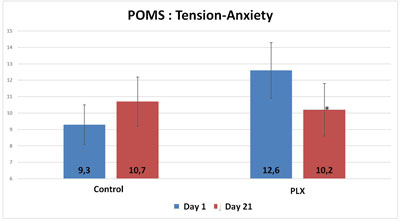
Thus, move!plx® appears to help reduce oxidative stress and muscle soreness, thereby contributing to the reduction in inflammation that in turn reduces muscle soreness and improves sleep and mood in athletes.To summarize, it appears that a reduction in inflammation (reduction in MPO activity) accompanied by a reduction in muscle soreness led the volunteers in the move!plx® group to have more positive feelings and higher POMS scores than the control group.
move!plx® is thus beneficial for sleep and mood.
3. move!plx® and joints
Reduction in pain and enhanced joint mobility
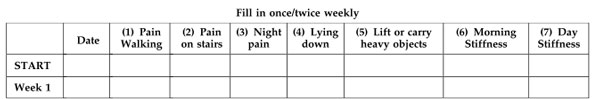
The fourth clinical study addressed joint mobility (Cartula et al., 2011). This controlled, randomized, double-blind study was carried out with a mixture of PLX® and omega-3 fatty acids. The number of participants was 12 in the placebo group (22 initially) and 19 in the PLX® group (23 initially). The administered dose was 230 mg at 14% verbascosides and 83.25 mg EPA/DHA (10/8) for 5 weeks, after which the dose was halved for 4 weeks. Participants filled out WOMAC and Lequesne questionnaires at the beginning of the study and then once a week.
The Western Ontario MacMaster (WOMAC) questionnaire is a validated index that assesses osteoarthritis in the lower limbs, as follows:
The Lequesne questionnaire is also called the Index of Severity for Osteoarthritis (1987) and assesses hip and knee osteoarthritis.
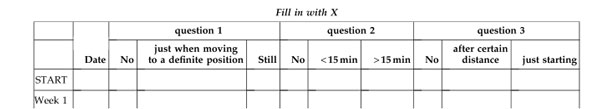
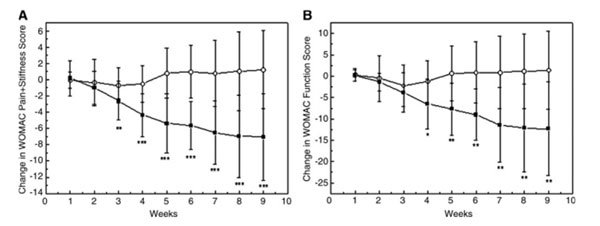
Figures A and B show WOMAC scores of pain/discomfort and function. The black squares represent the PLX® group and the white circles the control group. During the 9 weeks, there was a progressively stronger and significant decrease in the PLX® group than in the control group ( * p <0.05 **p <0.01 ***p <0.001 ).
The figure below shows the changes that occurred over 9 weeks in the Lequesne index, with the same pattern. There was a progressively stronger and significant decrease in the supplemented group compared with the control group. (Statistical analysis * p<0.05 **p<0.01).
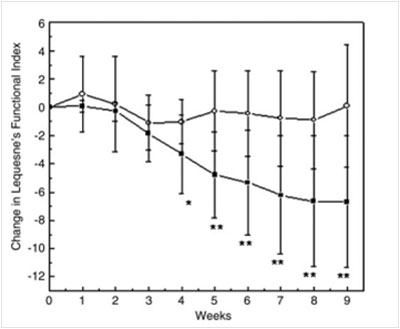
Cartula et al. observed a significant reduction in joint pain and a concomitant improvement in joint mobility.
Thus, PLX is beneficial for the well being of people and especially sportsmen.
Given the length of the study (9 weeks), this effect may explain the high rate of desertion of volunteers in the placebo group who did not notice any beneficial effects.
Sources
- Study 1: Carrera-Quintanar L., Funes L., Viudes E., et al. Antioxidant effect of lemon verbena extracts in lymphocytes of university students performing aerobic training program. Scand. J Med. Sci. Sports. 2012; 22 (4):454-61, 2010.
- Study 2: Funes L., Carrera-Quintanar L., Cerdan-Calero M. et al. Effect of lemon verbena supplementation on muscular damage markers, proinflammatory cytokines release and neutrophils’ oxidative stress in chronic exercise. Eur. J. Appl. Physiol. 2011; 111(4):695-705, 2010.
- Study 3: Martinez-Rodriguez A., Moya M., Vicente-Salar N. et al. Biochemical and psychological changes in university students performing aerobic exercise and consuming lemon verbena extracts. Current Topics in Nutraceutical Research, 2015.
- Study 4: Caturla N., Funes L., Pérez-Fons L. et al. A randomized, double-blinded, placebo-controlled study of the effect of a combination of lemon verbena extract and fish oil omega-3 fatty acid on joint management. J. Altern. Complement. Med.2011; 17 (11):1051-63, 2011.
move!plx® is an aqueous extract of lemon verbena (Aloysia citrodora Palau) that is listed in Annex I of the French decree on plants authorized for use in food supplements (“Plants” Decree of 24 June 2014). move!plx® is therefore authorized in food supplements.
Aloysia citrodora Palau is also listed in the “Novel Food Catalogue” under the names of Lippia triphylla or Lippia citrodora and is authorized in food and drink.
move!plx® is available as a water-soluble brown powder and benefits from two-year use date in its original packaging.
To benefit from the move!plx® effects on exercise recovery, the recommended dose is 400 mg of move!plx® at 30% verbascosides or 1200 mg of move!plx® at 10% verbascosides.

The only health claims that can be used for move!plx® are “health claims on plant-derived substances awaiting assessment” that specifically include lemon verbena.
There is 2 health claims “on hold” on the relaxing effect of move!plx® and physical well-being and joints.