
Lipophytol™ - Water-dispersible plant sterols for cholesterol reduction
Cardiovascular Health
Lipophytol™ is a micronized and microencapsulated source of phytosterols for cholesterol reduction.
Lipophytol™ is a water-dispersible powder with a phytosterol concentration of up to 90%. It also comes in liquid form. Lipophytol® lends itself to the development of innovative dietary supplement presentations and provides new ways of introducing phytosterols into the diet.
Lipophytol™ is safe, efficient and has potential cholesterol maintenance and cholesterol reduction properties.
Lipophytol™ is available in France and Benelux.
Due to their insolubility in water and lipids, enriching foods and beverages with phytosterols is very problematic.
Lipophytol™ is a dispersible form of phytosterols that have been microencapsulated to facilitate their incorporation into food matrices and dietary supplements. This unique delivery system increases phytosterol dispersibility in foods and beverages, thus providing a convenient way of introducing phytosterols into the human diet.
Lipophytol™ is made with pine tree sterols (Lipophytol™ P).
The main applications for Lipophytol™ are dairy products (milk, yogurt) and dietary supplements.
Phytosterols are currently very popular in dietary supplements due to recent safety doubts regarding red rice yeast sources. See Lipophytol™ Regulatory Information tab.
High blood cholesterol is the leading risk factor for coronary heart disease. Studies have shown that a 10% decrease in blood cholesterol levels can reduce the risk of cardiovascular disease by 19% to 54% depending on age.
The effect of plant sterols on cholesterol levels is well documented in the literature. Consumption of 1.5 to 3 g of plant sterols per day can lower LDL cholesterol by 8% to 15%.
Lipophytol™ efficacy
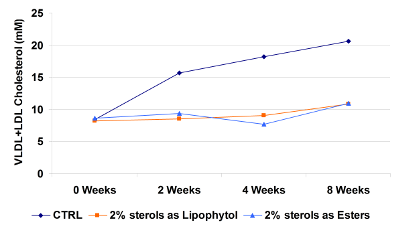
The effect of Lipophytol™ on cholesterol reduction was studied in vivo using knockout mice. Three groups of mice were fed either a high fat diet (control), a high fat diet with Lipophytol™ or a high fat diet with phytosterol esters.
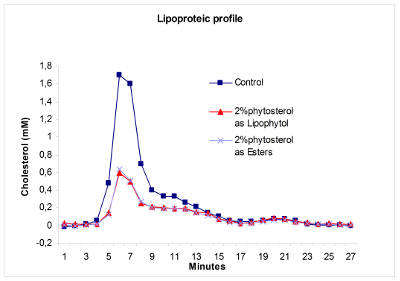
Lipophytol™ significantly reduced the concentration of VLDL and LDL cholesterol in the serum compared to the control.
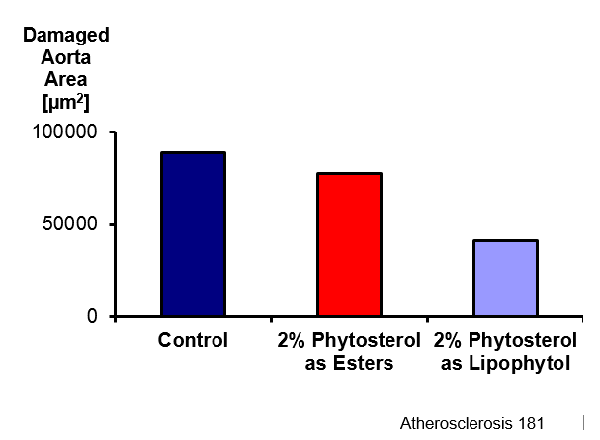
The effect of Lipophytol™ on aorta atherosclerotic lesions was examined in every group of mice after 8 weeks of treatment. Heart and proximal aortas were removed and artherosclerotic lesions quantified.
The Lipophytol™ group showed fewer damaged aorta compared to the control mice.
Reference: Dr. Blanco from the Danta Creu I San Pau Hospital in Barcelona (Spain)
Lipophytol™ was granted a ‘substantial equivalence’ approval by the Finnish Food Safety Authority.
In food supplements, phytosterols are not considered as novel ingredients as specified by Regulation (EC) No. 258/1997.
Italian authorities included phytosterols in the list of ingredients authorized in dietary supplements. (Website Ministero della Salute and the reglementation).
Labeling of food and beverage products containing phytosterols is subject to Commission Regulation (EC) No. 1169/2011, annexe II.

Lipophytol™ safety
In 2003, the Scientific Committee on Food (SCF) issued an opinion on the safety of phytosterols: “Provided that …enriched food products (with phytosterols) are not consumed in amounts resulting in total phytosterol intakes exceeding 3 g per day, the Committee concludes on the basis of the available toxicological and nutritional data on phytosterol esters and phytosterols in accordance with the previous opinions (SCF, 2000; SCF, 2002), that the use of phytosterols in these foods is safe.”
However, doubts were recently raised regarding the safety of red yeast rice in dietary supplements. The ANSM (French Drug Safety Agency) and ANSES (French Food Safety Authority) issued two opinions on the risks related to the consumption of red yeast rice in dietary supplements.
Lipophytol™ is therefore a good alternative to red yeast rice in dietary supplements.
Free (non-esterified) phytosterols are high meting point fats (145°C) that are insoluble in water as well as in oils. Their addition to food and beverages is therefore challenging for food and beverage formulators.
Lipophytol™ is a dispersible form of phytosterols that have been micronized and microencapsulated to facilitate their incorporation into food matrices and dietary supplements. This unique delivery system reduces particle size to below the perception threshold in the mouth (no grainy sensation) and increases phytosterol dispersibility in food and beverages, thus providing a convenient way of introducing phytosterols into the human diet.
In dairy products, Lipophytol™ should be mixed with milk using a Tri-blender® (or similar apparatus), homogenized and heat-treated or spray-dried. Lipophytol™ withstands temperatures typical of pasteurization and UHT treatments.
Homogeneization is needed to disperse microcapsules stuck together during the drying process of Lipophytol™. Only Lipophytol™ Liquid does not require any homogeneization treatment to reduce microcapsule size below the perception threshold.
- Lipophytol™ P is contains up tp 90% phytosterols and can be used in dietary supplements presented in powdered or tablet form.
- Lipophytol™ ME is a granulated form, contains 70% phytosterols and is very compressible. Il can be used in tablets.
- Neither Lipophytol™ P or ME can be used in hard gel capsules.
- Lipophytol™ Liquid is a dispersion of micronised phytosterols in water and contains a minimum of 25% phytosterols. It is a very innovative presentation of phytosterols. Lipophytol™ Liquid can be used in any liquid dietary supplement, beverage or food.
The European Commission has approved two health claims for plant sterols, namely:
- Plant sterols/stanols contribute to the maintenance of normal blood cholesterol levels (Commission Regulation (EU) No. 432/2012)
- Plant sterols and plant stanol esters have been shown to lower/reduce blood cholesterol. High cholesterol is a risk factor in the development of coronary heart disease (Commission Regulation (EU) No. 384/2010).
No. 983/2009 : Health claims
No. 384/2010 : Adding and denial claims about No. 983/2009
No. 686/2014 : Modification No 983/2009 and No. 384/2010
