
Powdered fruit sugars
Energy, diabetes
Naturalia’s ingredients natural grape sugar extracts: fructose, dextrose (D-Glucose) and mixtures. These natural healthy ingredients exist in liquid and powder form.
The fructose/dextrose mixture is available with an organic certification. ![]()
Fruit sugars are available in France.
Naturalia fruit sugars are naturally present in grapes:
- Crystal GrapeSugar®: natural mixture of grape fructose and dextrose
- Crystal FructoGrape®: natural fructose from grapes
- Crystal DextroGrape®: natural dextrose from grapes
Only Naturalia fruit sugars are available in crystallised form, offering a wide range of new applications.

Crystal DextroGrape®: The D-Glucose monohydrate is a natural sugar exclusively available in powder form. It is at least 99.5% pure and provides sweetening power of between 50% and 70% of saccharose.
Crystal FructoGrape®: This natural sugar is available in powder form at 98% purity or in liquid form with different Brix degrees. Its sweetening power is between 1.3 and 1.5 times higher than saccharose.
Crystal GrapeSugar®: Composed of 50% dextrose and 50% fructose. It is available in powder and liquid form at different Brix degrees. Its sweetening power is close to that of saccharose.
1. Fructose from fruit reduces postprandial glycaemia
Postprandial glycaemia is the increase in blood glucose levels after food intake. This is a normal physiologic response, which varies in intensity and duration. Glycaemia can be influenced by the physicochemical nature of the food and other individual factors. Decreasing the postprandial glycaemic response may be beneficial for glucose intolerant people (Venn and Vert, 2007).
Several studies have assessed the effects of fructose on glycaemic and insulinemic postprandial responses in comparison to other carbohydrates.
A study was carried out on 36 people: 9 healthy people, 10 glucose intolerant people and 17 type-2 diabetic people. In a randomised order they received 50g of glucose, 50g of saccharose and 50g of fructose. Blood samples were taken 0, 15, 30, 45, 60, 120 and 180 minutes after each meal.
It showed that fructose consumption caused a significantly lower glycaemic and insulinemic response than glucose or saccharose consumption in all groups. Moreover, the glycaemic postprandial response was proportional to the level of glucose intolerance. (Crapo et al. (1980))

Another study was carried out on 32 people: 10 healthy people, 12 type-1 diabetic people and 10 type-2 diabetic people without oral blood glucose-lowering agent intake. In a randomised order the participants received five test breakfasts containing between 42g and 43g of different carbohydrates (glucose, fructose, saccharose, potato starch and wheat starch). Their energy intake was almost identical (685-742 kcal), as well as total content of carbohydrates (84g to 89g), proteins (31g to 38g) and lipids (25g to 26g). Blood samples were taken 0, 15, 30, 60, 90, 120, 180 and 240 minutes after each breakfast.
The breakfast with fructose reduced postprandial glycaemia in comparison with the one containing glucose in healthy and type-2 diabetic people. The responses to insulin of the different breakfasts did not differ (Bantle et al, 1983).

The third study comparing fructose and the effects of other carbohydrates (glucose, saccharose, etc.) was conducted on 10 people with type-2 diabetes. Consumption of fructose instead of other carbohydrates over a 3-week period reduced their postprandial glycaemia (Koivisto, Yki-Jarvinen (1993)).
Finally, a study was conducted in 2001 on 9 healthy subjects, 10 glucose intolerant people and 17 patients with type-2 diabetes. It also showed that the diabetic patients who received fructose had a lower glycaemia than people who consumed glucose at the end of the study (Ionescu-Tirgoviste C, (2001)).
Moderate fructose consumption reduces postprandial glycaemia and blood insulin levels compared to glucose, saccharose and other carbohydrates. This effect is supported by a health claim granted by the European Commission in 2013.
However, several intervention studies showed that excessive fructose consumption (particularly in sodas) increased the risk of dyslipidemia, insulin resistance and increased levels of visceral fat (Review in Le and Tappy, (2006, 2010); Stanhope and Havel, (2008, 2009, 2010)).
These effects were not observed with recommended doses (40g to 50g per day as a substitute for starch or saccharose (Wolever (2006)).
French regulations
Fruit sugars are defined as sugar under French Decree no. 2013-1049 of 21 November 2013.
European regulations
Crystal DextroGrape and Crystal FructoGrape are natural sugars and comply with Directive 2001/111/EC. Purified and crystallised D-Glucose, which contains a crystallisation water molecule, has to comply with the following features:
- Dextrose (D-glucose): not less than 99.5% by dry weight
- Dry matter: not less than 90% by weight
- Sulphated ashes: no more than 0.25g/100g dry matter
Purified and crystallised Fructose has to comply with the following requirements:
- Fructose: not less than 98%
- Glucose: not more than 0.5%
- Loss in drying: not more than 0.5%
Both products comply with the Codex Alimentarius and the Food Chemical Codex.

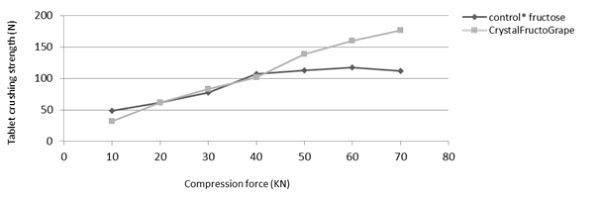
1. Crystal FructoGrape: compressible but hydroscopic
Natural fruit sugars can be used as sweetener, bulking agent, compacting agent or for technological uses (conservation, texturing, anti-crystallising). They can be found in capsules, oral solutions, syrups, etc.
Crystal FructoGrape: compressible but hydroscopic
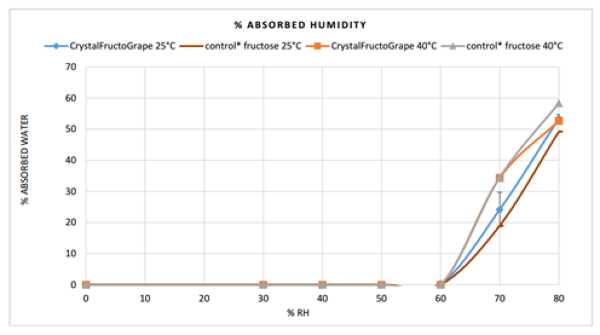
Crystal FructoGrape is an excellent compressing agent. However, it must be maintained at 25°C maximum and in an environment with humidity below 60%.
Compression graphs:

Crystal FructoGrape sorption isotherm:
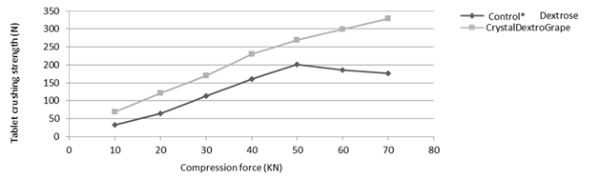
Crystal DextroGrape
Compression graphs
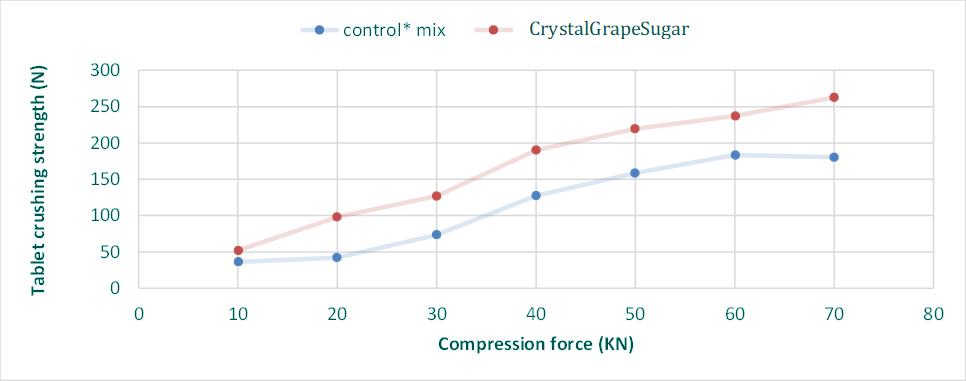
Crystal GrapeSugar
Compression graphs
The European Commission authorised a health claim for the use of fructose in Regulation (EU) no. 536/2013.
The health claim is as follows: “Consumption of foods containing fructose leads to a lower blood glucose rise compared to foods containing sucrose or glucose”.
Fructose can be used as a replacement for glucose and/or sucrose in sugar-sweetened foods or drinks to bring about a reduction in the glucose and/or sucrose content of these foods or drinks of at least 30%.
