Lipofer™ - A tasteless, non-pro-oxidative source of iron
Fatigue, Cognitive Health and the Immune System
Lipofer™ is a micronized and microencapsulated source of iron that does not taste metallic or oxidize unsaturated fats. Claims on cognitive function or development (Article 14), tiredness, energy and immune system can be made for Lipofer™.
Lipofer™ is available in France and Benelux.
Supplementing food matrices with iron is plagued by numerous organoleptic problems (metallic taste), technological problems (oxidation of fats and vitamins) and nutritional problems (poor bioavailability).

Lipofer™ is a microencapsulated and micronized source of iron that overcomes these problems without adverse effects.
1. No metallic taste
- The most common problem in iron enrichment is an unpleasant metallic taste. However, the microencapsulation technology used in Lipofer™ offsets the characteristic metallic taste of iron solutions.
2. No oxidation of fats and vitamins
- Its unique microencapsulation delivery system inhibits the interaction with other food ingredients. Thus, highly sensitive food ingredients such as vitamins or unsaturated fatty acids are protected against the oxidative capacity of free iron. The final product is stable, preserving its nutritional and sensory qualities throughout its entire shelf life.
3. Easy to use
- Lipofer™ is available as a water-dispersible powder standardized at 8% elemental iron. The powder form facilitates its use in many kinds of foods and beverages and can be easily added during the manufacturing process. It is stable in acidic conditions and can withstand high temperatures (stable at 121ºC for 20 min) and pH as low as 2.5.
1. Iron deficiency: effects on health
According to the World Health Organization (WHO, 2001), iron deficiency, especially iron deficiency anemia, remains the main severe nutritional disorder in the world today. Also affecting women and children in non-industrialized countries, iron deficiency is the only nutrient disorder that is widespread in all industrialised nations.
However, adverse functional consequences can occur even before anemia sets in. The increased fatigue and weakness, which leads to a reduced work capacity, is well-known (Haas & Brownlie, 2001). In addition, impairment of the immune system and cognitive development of infants are also linked to iron deficiency.
2. Immune System
Morbidity (affinity to contract diseases) from infectious disease is increased in iron-deficient populations. The immune system is impaired by iron deficiency because leukocytes have a reduced capacity to kill ingested microorganisms and B-lymphocytes have a decreased ability to replicate when stimulated by pathogens.In such cases, a lowered concentration of cells responsible for cell-mediated immunity occurs (Beard, 2001; Brock & Mulero, 2000).
In addition, different studies reviewed by the WHO have demonstrated a strong relationship between iron deficiency and an impaired immune system. For example, iron supplementation in iron-deficient children reduces morbidity from infectious diseases (WHO, 2001).

3. Physical performance and work productivity
In developing countries where people often work as farmers, physical work capacity is of particular importance for family well-being. According to the WHO (2001), physical work capacity and performance is impaired by up to 30% in iron-deficient men and women. Many studies have shown that work capacity is considerably improved after iron supplementation (Beard, 2001).
4. Cognitive development
It is well known that anemia during infancy and childhood can severely affect a child’s cognitive development (Beard, 2003). Young anemic children will have great deficit in IQ compared with their classmates without anemia and they will show impaired performance in almost all tested skills (Lozoff, 1989).
Since later treatments are considered ineffective, it is essential that iron supplementation of deficient infants and children is administered as early as possible (WHO, 2001, Halterman et al., 2001). Not only anemia but also iron deficiency can impair cognitive functioning. In contrast to anemia, this effect seems to be able to be corrected by iron administration (Bruner et al. 1996).

5. Anemia
If iron deficiency is not treated for a long time, anemia can develop. About 25% of the world’s population is estimated to be anemic (WHO, 2008). Even in developed countries, the prevalence of anemia can be considered as relatively high: in Europe the prevalence of anemia among preschool children is 16.7% (WHO, 2008).
During pregnancy, anemia is associated with multiple adverse outcomes for both mother and infant, including an increased risk of hemorrhage, sepsis (blood stream infection), maternal mortality, perinatal mortality, and low birth weight (Scholl & Hediger, 1994). The risk of maternal mortality is higher when there is iron-deficiency anemia (International Nutritional Anemia Consultative Group INACG, 2002).
6. Recommended Daily Allowance (RDI) and Population Requirements Intake(PRI)
Nutritional requirements depend on the specific needs of individuals, especially for iron, which in turn depend on sex, age and physiological status (pregnancy, breastfeeding women), etc. Based on the population reference intake (PRI) values published by the Scientific Committee for Food (SCF, 1992 and 1993), the European Union set the recommended daily allowance (RDA) for iron to 14 mg/day (Annex to Directive 90/496/EEC amended by Commission Directive 2008/100/EC).
Annex XIII of Regulation No. 1169/2011 on the provision of food information to consumers (that comes into effect by 13 December 2014) replaces the term RDA with “Nutrient Reference Values” (NRVs) without changing any nutrient values.
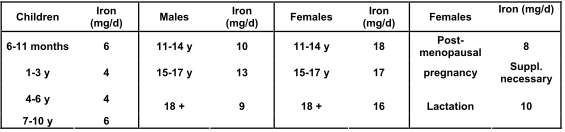
PRI values established in 1992 for iron by the SCF for the EU: intakes of iron must cover the requirements of 95% of the various population groups, based on a biovailability of 15%.
7. Iron Deficiency in Europe
The European Nutrition and Health Report of 2004 (Elmadfa et al., 2004) showed an insufficient iron intake in different groups of the population in almost all European countries.
a. Babies and children
Infants have relatively high iron requirements because they are growing very rapidly. Infants are normally born with plenty of iron. However, after 6 months of age, the iron content of maternal milk is sometimes insufficient to meet an infant’s requirements. Low birth weight infants (less than 2500 g) are born with lower iron stores and are at high risk of deficiency as soon as they are 2 months old (WHO, 2001). Where iron-fortified complementary foods are not widely and regularly consumed by young children, infants should routinely receive iron supplements in the first year of life (WHO, 2001).

b. Female adolescents & women of childbearing age
Iron deficiency and the more severe iron-deficiency anemia frequently occur in male and female adolescents due to accelerated growth, rapid increase in blood volume and muscle mass. Additionally, female adolescents often do not consume sufficient iron to offset menstrual losses. As a result, a peak in the prevalence of iron deficiency frequently occurs among females during adolescence (WHO, 2001). The European Nutrition and Health Report of 2004 shows that especially girls at fertile age in all European countries do not absorb enough iron with their daily diet.

Moreover in industrialized countries, women tend to limit their food intake to control their weight. Slimming diets or poor eating habits lead teenage girls or women of childbearing age result in low iron intake.
The intra-uterine device is an additional risk factor because it can double mentrual blood volume whereas the pill is known to reduce menstrual blood volume.
According to the WHO, 10.3% of women (15-59 years) living in industrialized countries have anemia, and 42.3% in non-industrialized countries. In its 2001 report, WHO estimates that the prevalence of iron deficiency is two to five times higher than the prevalence of iron-deficiency anemia. This implies that 20% to 50% of women living in industrialized countries and virtually all women in developing countries suffer from iron deficiency.
The European Nutrition and Health Report of 2004 (Elmadfa et al.) states that all women of childbearing age and older have an iron intake that is not sufficient to fulfill their needs.
c. Female and male athletes
Among the athletes, the prevalence of iron depletion without anemia seems to be significantly higher than in the general population. Endurance athletes are particularly prone to iron deficiency. The reasons for this iron depletion is footstrike hemolysis, poor dietary habits, increased iron losses through sweat, fecal blood losses and desquamated epithelial cells (Akabas & Dolins, 2005 ; Sinclair & Hinton, 2005). As mentioned above, iron is needed for energy production. The iron-containing heme group of hemoglobin transports oxygen from the blood to the muscles where oxygen is stored in myoglobin which also contains heme iron. Moreover, in the mitochondria, iron is an essential cofactor of the iron-dependent respiratory chain. When mitochondria are not supplied with enough iron, energy production is impaired. The result is an impact on physical performance which first affects athletes (Beard, 2001; Dubnov & Constantini, 2004).

8. Lipofer™ efficacy
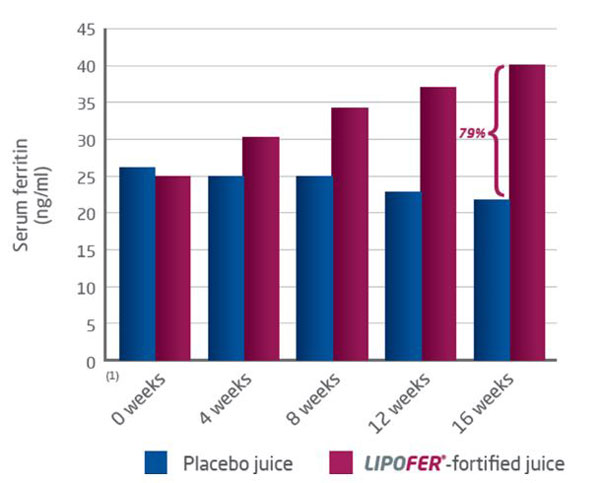
Lipofer™-fortified fruit juice improves iron status in menstruating women with low iron stores.
A double-blind study controlled by placebo investigated the influence of consuming fruit juice fortified with Lipofer™ on iron status in 130 menstruating women, aged 18 to 35 years, with low iron stores.
Subjects were randomized into a placebo group and a fortified group who, as a supplement to their usual diet, consumed 500 ml per day of a placebo fruit juice or a juice fortified with Lipofer™, respectively, for 16 weeks. The fortified juice provided 16 mg of elemental iron.
Monthly blood samples were collected from the study participants and the concentration of serum ferritin was determined. by a Modular Analytics Serum Work Area analyser (Roche, Basel, Switzerland)
Lipofer™-fortified fruit juice consumption significantly improved the iron status in the fortified group compared to the control group: ferritin was 79% higher than in the control group after 16 weeks. (p<=0.001).
Blanco-Rojo, R. et al. 2011. Efficacy of a microencapsulated iron pyrophosphate-fortified fruit juice: a randomised, double-blind, placebo-controlled study in Spanish iron-deficient women. British Journal of Nutrition vol.105:1652-91.
Lipofer™ NA Dispersable can be used in :
- foods as allowed by Regulation (EC) No. 1925/2006 on the addition of vitamins and minerals and of certain other substances to foods;
- foods for particular nutritional uses as allowed by Commission Regulation (EC) No. 953/2009 on substances that may be added for specific nutritional purposes in foods for particular nutritional uses;
- food supplements as allowed by Directive 2002/46/EC on the approximation of the laws of the Member States relating to food supplements;
- infant formulae as allowed by Commission Directive 2006/141/EC on infant formulae and follow-on formulae and amending Directive 1999/21/EC;
- cereals and baby foods as allowed by Commission Directive 2006/125/EC on processed cereal-based foods and baby foods for infants and young children.
Annex XIII of Regulation No. 1169/2011 on the provision of food information to consumers, the significant amount of iron is :
- 15 % of the nutrient reference values specified in point 1 supplied by 100 g or 100 ml in the case of products other than beverages,
- 7,5 % of the nutrient reference values specified in point 1 supplied by 100 ml in the case of beverages,
- 15 % of the nutrient reference values specified in point 1 per portion if the package contains only a single portion,
Supplementing food matrices with iron is plagued by numerous organoleptic problems (metallic taste) and technological problems (oxidation of fats and vitamins).
The microencapsulation technology used in Lipofer™ offsets the characteristic metallic taste of iron solutions and inhibits the interaction with other food ingredients. Thus, highly sensitive food ingredients such as vitamins or unsaturated fatty acids are protected against the oxidative capacity of free iron.
Lipofer® is available as a water-dispersible powder standardised at 8% elemental iron. The powder form facilitates its use in many kinds of foods and beverages and can be easily added during the manufacturing process of dairy products.
Lipofer® can be used in liquid and powdered milk:
- Pasteurized and UHT milk
- Evaporated milk: Lipofer can withstand 121ºC for 20 min
- Spray-dried milk and infant milk

Lipofer® can be used in fermented milk products such as yogurt. It is stable in acidic conditions with lactic acid at pH as low as 2.5.
Lipofer® should not be used in fruit preparations or juices where anthocianins, citric acid and malic acids may form iron complexes and cause a change in color and flavor of the food or drink.
Lipofer® is used in infant formulae or in dietary supplements together with DHA to avoid oxidation of DHA. Stability trials need to be done to validate the stability of PUFAs with LIPOFER®.
Nutrition Claims
According to the Corrigendum to Regulation (EC) No. 1924/2006 and the Annex to Directive 90/496/EEC amended by Commission Directive 2008/100/EC, foods or beverages with added iron can be labelled
- “source of iron” when they contain at least 15% of the recommended daily allowance (RDA) per 100 g, 100 ml or per portion
- “high in iron” when they contain at least 30% of the RDA per 100 g, 100 ml or per portion.
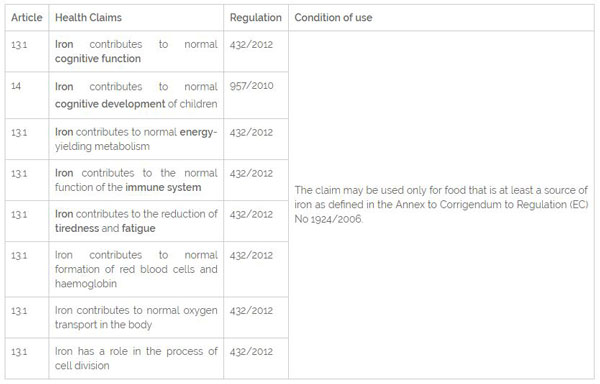
Health Claims